
With over 600 thousand new cases and more than half that would lead to death each year, cervical cancer is listed as the fourth most frequent cancer among women worldwide. According to WHO, a subset of HPV (Human Papillomavirus) types is responsible for almost all cases of cervical cancer, 70% of which is highly correlated with both HPV 16 and 18 the most oncogenic types worldwide. Lack of high-quality cervical cancer screening and treatment leads to higher rates of cervical cancer incidence and mortality, especially in low- and middle- income countries.
- Double-strand DNA virus
- Only infect with human beings
- 100+ type founded with 40+ infected with genital tract
- High-risk: carcinogenic genital HPV causing cervical cancer and others (anus, vulva, vagina, penis, and oropharynx cancer)
Based on this acknowledgment, the WHO has established the well-known global strategy to eliminate cervical cancer worldwide in November 2020. The clear goal setting for the world is that by 2030, cervical cancer shall be eliminated targeting the incidence rate of fewer than 4 new cases of per 100 thousand women per year.
In order to meet the targets step by step, the following 90-70-90 interim targets have been put forward based on the mathematical model illustration that the incidence and mortality rates will gradually decrease attributed to the wide-scale implementation of population-based screening and treatment, and vaccination.
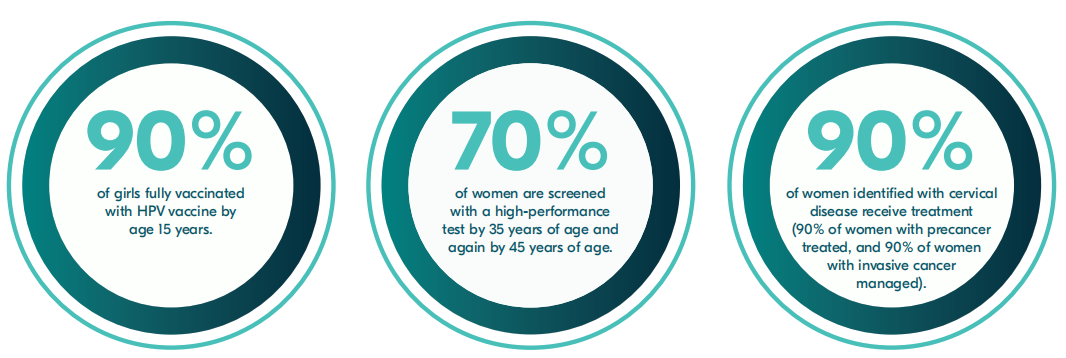
WHO highly recommends that HPV-DNA test could be taken as a primary screening test with proven superiority such as cost-effective, requiring fewer trained expert pathologists or cytotechnologists. Many programmes including political cooperation are transitioning to HPV-DNA test in order to achieve the 2030 target as cervical cancer is curable if detected early and properly treated.
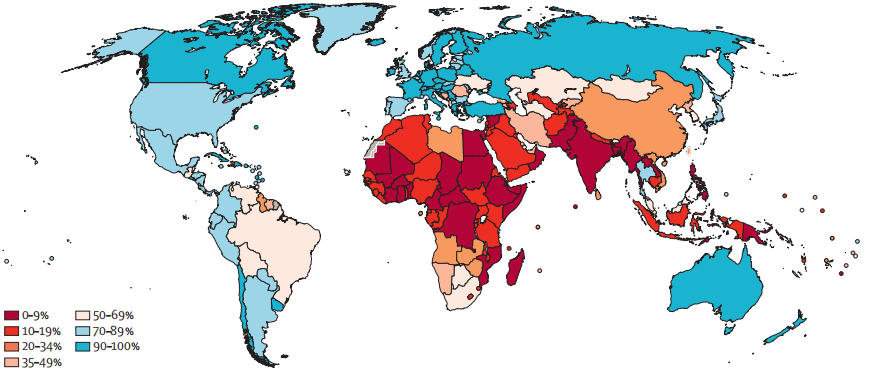
Ever in lifetime cervical cancer screening coverage in women aged 30–49 years in 2019 by country
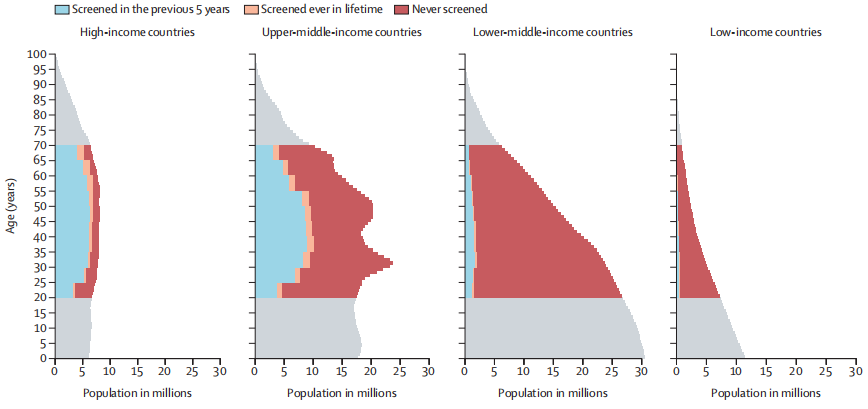
Female population pyramid by cervical cancer screening status and income level in 2019
The number of women is from the 2019 UN population estimates.
Countries are grouped according to the 2019 World Bank’s classification.
Illustrated data indicate a great demand for increasing screening and diagnostic capacity to meet the cervical cancer elimination target.
Bioperfuctus, a one-stop supplier, specializing in molecular diagnostic, highly recommends the total HPV PCR solution with higher throughput, higher efficiency, and more cover up 21 types for HPV detection: Human Papillomavirus Real Time PCR Kit and Human Papillomavirus Genotyping Real Time PCR Kits applied in the combination of SAW-96 Automated Nucleic Acid Extraction Workstation and STC-96A Plus Real-time PCR System.
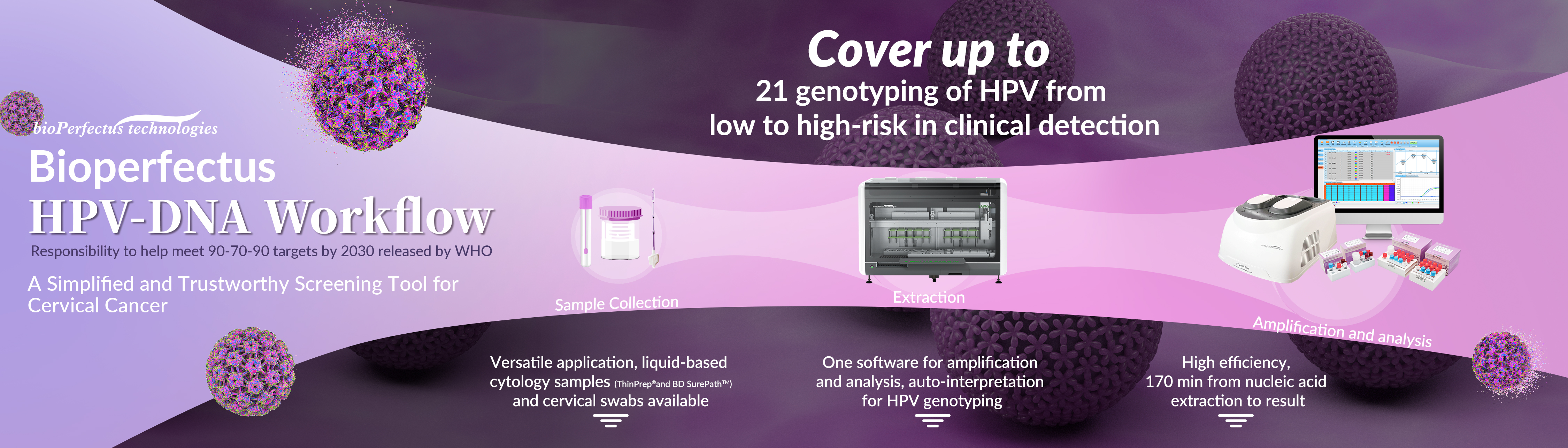
The total HPV PCR solution can detect liquid-based cytology specimens and cervical swabs. It supports auto operations of sample loading from primary/ secondary tubes, nucleic acid purification, and PCR setup with the assistance of SAW-96, which boasts the advantages such as outstanding efficiency, real-time visual interface, better contamination control, and so on. Besides, it provides the final sample results directly through the auto-interpretation function on a compatible STC software system.
Supported by one solution from one supplier, Bioperfectus is confident to provide partners and customers solid results around the world with a simplified and trustworthy HPV-DNA workflow for cervical cancer screening.
The Total HPV PCR Solution enables women to access screening and diagnosis at the early stage of HPV infection with accurate genotype detection and to get proper treatment before the development of precancerous lesions and cervical cancer. Pioneering in molecular diagnostic, Bioperfectus is always determined to pursue high quality, keep innovating and strive to solve health problems faced by mankind for a healthier future.
Part of International published articles by using Bioperfectus HPV Kits:
- Xiang T, Huina Z, Hao Z, Yaoxing X, Fangfang Z, Xianrong Z, Qing C, Long S Chengquan Z. The clinical utility of extended high-risk HPV genotyping in risk-stratifying women with L-SIL cytology: A retrospective study of 8726 cases. Cancer Cytopathology 21 March 2022 doi: 10.1002/cncy.22573. Available at: https://acsjournals.onlinelibrary.wiley.com/doi/abs/10.1002/cncy.22573
- Tao X, Zhang H, Wang S, Chen T, Cong Q, Wang L, Zhou X, Zhao C. Prevalence and carcinogenic risk of high-risk human papillomavirus subtypes in different cervical cytology: a study of 124,251 cases from the largest academic center in China. J Am Soc Cytopathol. 2021 Jul-Aug;10(4):391-398. doi: 10.1016/j.jasc.2021.03.006. Available at: https://www.sciencedirect.com/science/article/abs/pii/S2213294521000417
- Chen L, Dong B, Zhang Q, Mao X, Lin W, Ruan G, Kang Y, Sun P. HR-HPV viral load quality detection provide more accurate prediction for residual lesions after treatment: a prospective cohort study in patients with high-grade squamous lesions or worse. Med Oncol. 2020 Mar 30;37(5):37. doi: 10.1007/s12032-020-01363-z. Available at: https://link.springer.com/article/10.1007%2Fs12032-020-01363-z
Reference:
-Laia Bruni, et al. Cervical cancer screening programmes and age-specific coverage estimates for 202 countries and territories worldwide: a review and synthetic analysis, The Lancet Global Health, Volume 10, Issue 8, 2022, Pages e1115-e1127.
-WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention, 2nd edition
-Accelerating the elimination of cervical cancer as a public health problem: Towards achieving 90–70–90 targets by 2030
-https://www.who.int/news-room/fact-sheets/detail/cervical-cancer

