
Today, we Bioperfectus are thrilled to announce that our Novel Corona Virus (SARS-CoV-2) Ag Rapid Test Kit (self-test) has received the approval ofBfArM. This remarks another milestone after we passed the PEI evaluation. It means that we are allowed to sell the self-test kit inGermanyfor direct pathogen detection of coronavirus SARS-CoV-2.
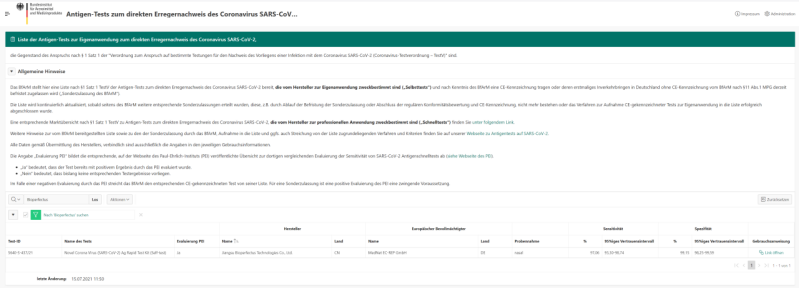
Click the picture above to find “Bioperfectus” in the list
In the case of a special approval of antigen tests for self-testing, BfArM evaluates not only the clinical detection quality including a positive evaluation by the Paul Ehrlich Institute (PEI), but in particular the usability of the sample collection and test handling, including the instructions for use, which are specially designed for lay use.
Bioperfectus Novel Corona Virus (SARS-CoV-2) Ag Rapid Test Kit (self-test) passed the PEI assessment withclinical sensitivity 97.1% and clinical specificity 99.2%. With the flexible packages of 1T/2T/5T/10T/15T, Bioperfectus could meet the various test demands for self-testing. The Bioperfectus Novel Corona Virus (SARS-CoV-2) Ag Rapid Test Kit (self-test) has been verified to be SARS-CoV-2 mutation free and has received the positive result on asymptomatic patient samples from a CLIA certified laboratory. As one of the leading professional manufacturers of infectious disease diagnostic solution, we Bioperfectus are looking forward to bringing our high-quality and user-friendly self-test kit to your home.
References
- https://www.cms.gov/Regulations-and-Guidance/Legislation/CLIA

