- As one of the listed Covid-19 antigen rapid test kits that are recommended and authorized for use by the Ministry of Health, Brunei Darussalam, Bioperfectus SARS-CoV-2 Ag Rapid Test Kits were sent to Brunei in 3 batches.
- Upon the request of the Ministry of Health, Brunei Darussalam, Bioperfectus will supply the SARS-CoV-2 Ag Rapid Test Kits with the customized QR code cassettes, which utilizes the tracing & tracking ability of the COVID-19 cases in Brunei.
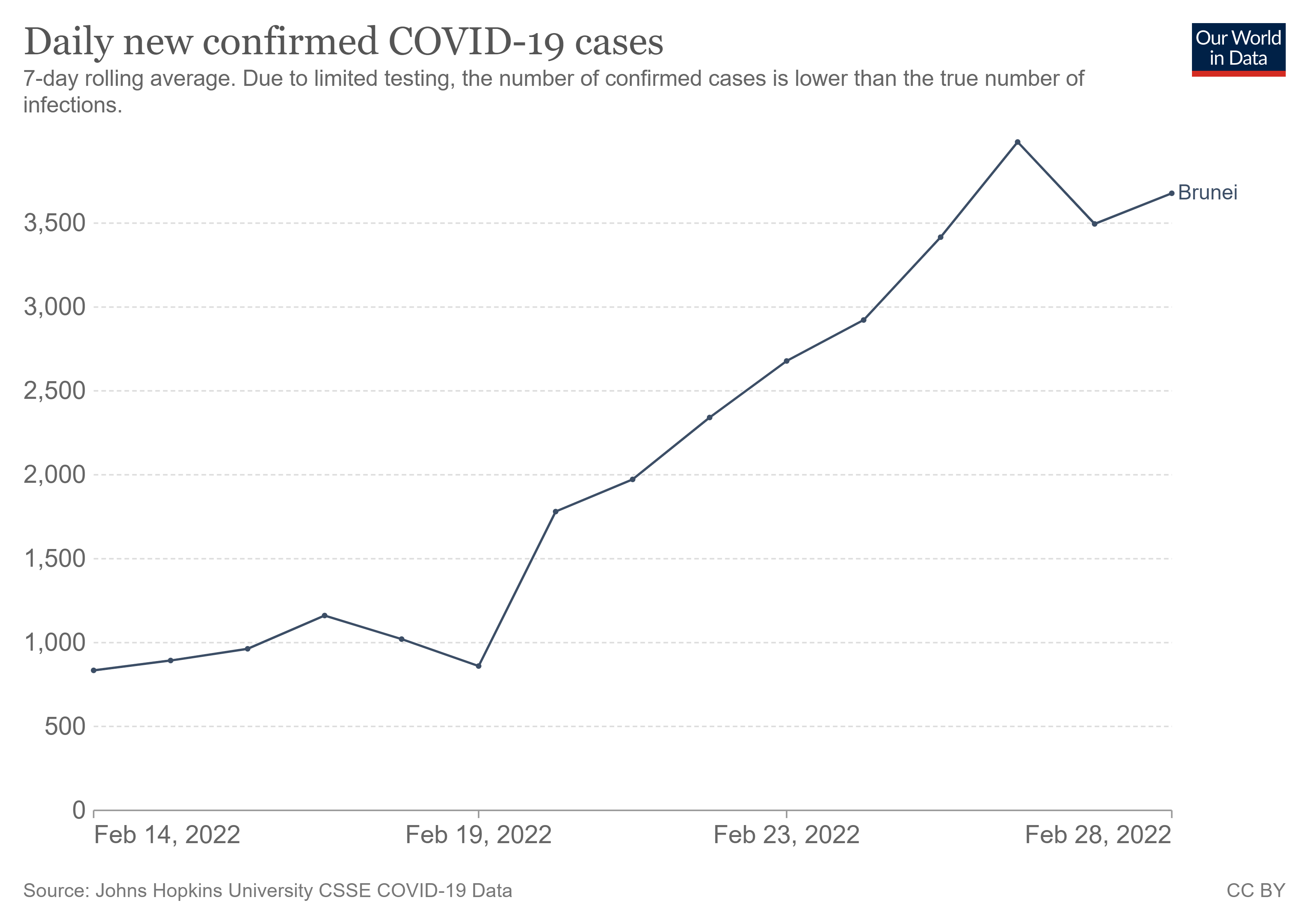
According to the latest COVID-19 cases report, as of March 1, 2022, there are a total of 59,447 COVID-19 cases outbroke in Brunei, for the last 7 days, there is an obvious surge in the local record.
 Since December 1, 2021, The Ministry of Health, Brunei Darussalam has appointed 12 clinics and healthcare businesses to issue certificates as proof of individuals taking COVID antigen rapid tests (ART).
Since December 1, 2021, The Ministry of Health, Brunei Darussalam has appointed 12 clinics and healthcare businesses to issue certificates as proof of individuals taking COVID antigen rapid tests (ART).
Bioperfectus SARS-CoV-2 Ag Rapid Test Kits were sent to Brunei in 3 batches after winning the bidding of the Ministry of Health, Brunei Darussalam. Bioperfectus SARS-CoV-2 Ag Rapid Test Kits are recommended and authorized for use based on the evaluation done by the Ministry of Health, Brunei Darussalam.

Upon the request of the Ministry of Health, Brunei Darussalam, Bioperfectus will supply the SARS-CoV-2 Ag Rapid Test Kits with the customized QR code cassettes, which utilizes the tracing & tracking ability of the COVID-19 cases in Brunei.

Together, we will fight and defeat COVID-19.

