
The ambitious goal of Eliminating Cervical Cancer Globally remains a significant focus, as does expanding these efforts to all cancers caused by HPV among women and men. In recent years, due to early screening and the improvement of public health awareness, the incidence of cervical cancer has dropped significantly compared with before. However, the five-year survival rate of patients in the middle and advanced stages is still around 55%.

EUROGIN 2022, the first large international in-person conference on HPV-related diseases. The theme of EUROGIN 2022 is “Accelerating detection and elimination of HPV and related cancer.” As the Bronze Sponsor of the upcoming EUROGIN 2022, Bioperfectus is glad to invite you to meet us in Düsseldorf, Germany, from April 10 to 12, 2022.
Venue: Congress Center, Düsseldorf, Germany
Date: April 10-12, 2022
Booth Number: #A01
Bioperfectus Products at the Upcoming EUROGIN 2022
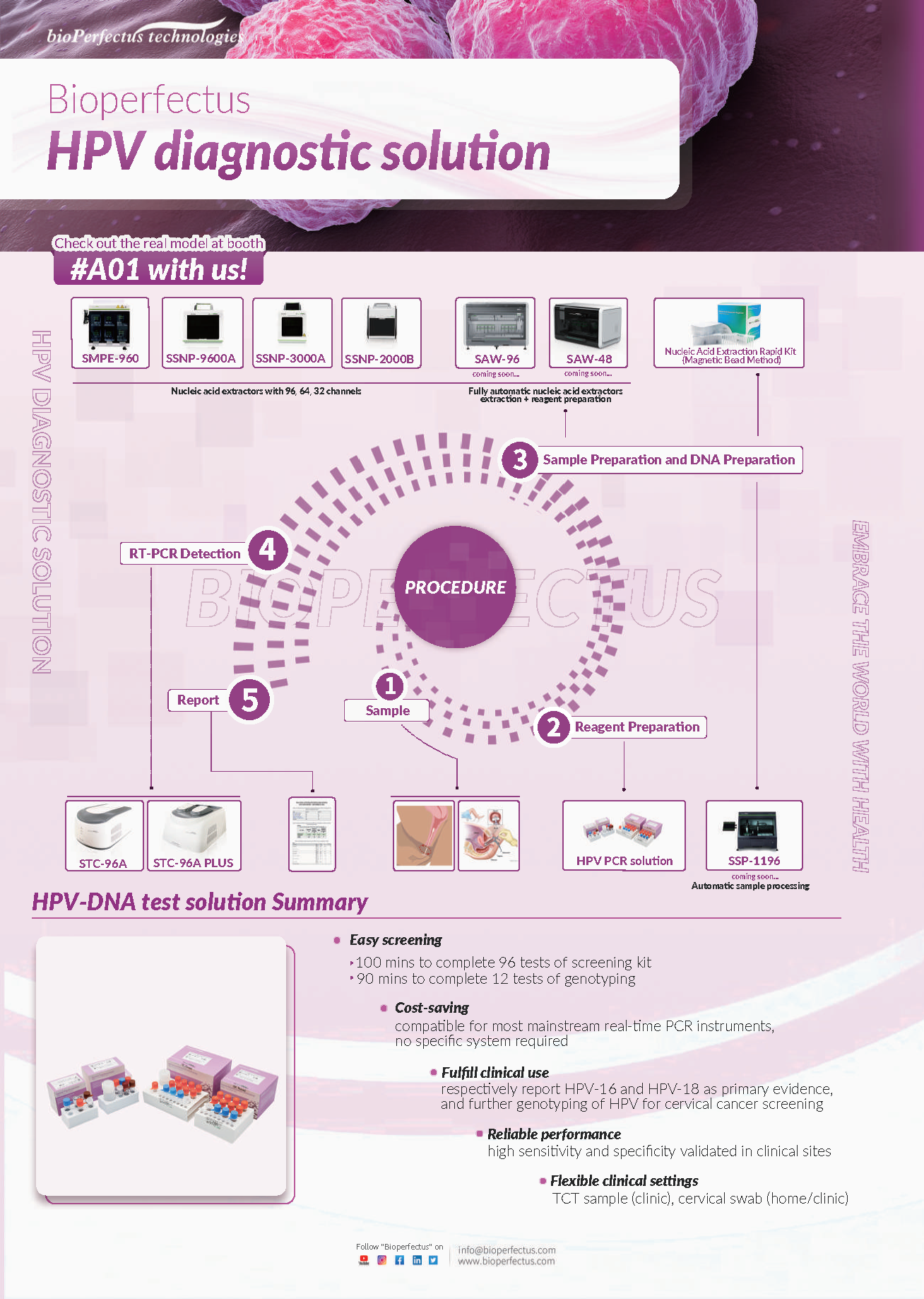
With the switch from cytology to HPV testing, Bioperfectus is dedicated to providing the fast, accurate, holistic HPV PCR total solution.
At EUROGIN 2022 this year, we Bioperfectus will present the products ranging from Human Papillomavirus Real-Time PCR Kit, Human Papillomavirus Genotyping Real-Time PCR Kit, Neisseria Gonorrhoeae/Chlamydia Trachomatis/U. Urealyticum Real-Time PCR Kit, Ureaplasma Urealyticum Real-Time PCR Kit, Chlamydia Trachomatis Real-Time PCR Kit, Neisseria Gonorrhoeae Real-Time PCR Kit, Nucleic Acid Extraction Rapid Kit (Magnetic Bead Method), Nucleic Acid Extraction System SMPE-960, to STC-96A PLUS Real-Time PCR System, etc.
To the Girls: Caring for Every Woman's Health
One Solution, One Supplier, One Response. If you want any further information regarding Bioperfectus HPV Testing Total Solution, please contact us via info@bioperfectus.com or marketing_global@bioperfectus.com
To learn more about the upcoming EUROGIN 2022, please click here.

