
Chlamydia is a common STD (Sexually Transmitted Disease) that can cause infection among men and women, leading to permanent damage to women’s reproductive system, making it difficult or impossible to get pregnant later. Even in the face of a pandemic, STD detection remains significant in public, and annual screening is recommended by CDC.
Section of STD/AIDS Control and Prevention, Jiangsu Provincial Center for Disease Control and Prevention, Nanjing, China, published their research on the High prevalence of Chlamydia trachomatis infection among women attending STD and gynecology clinics in Jiangsu province, China (A cross-sectional survey). The paper was published on Nov 19, 2021, in the Medicine (IF: 1.889).
The study collected cervical swabs from STD and gynecology clinics throughout Jiangsu province from 2018 to 2019. A total of 2664 female participants with urinary or genital tract symptoms, aged 18 to 60, were enrolled. It aimed to discover the factors which significantly affect CT infection rate.
441 participants (16.6%) tested positive for CT infection via Bioperfectus reagents in this cross-sectional survey. A significant correlation existed between CT infection and sub-district, occupations, and clinics. At the same time, there was no significant difference between age groups.
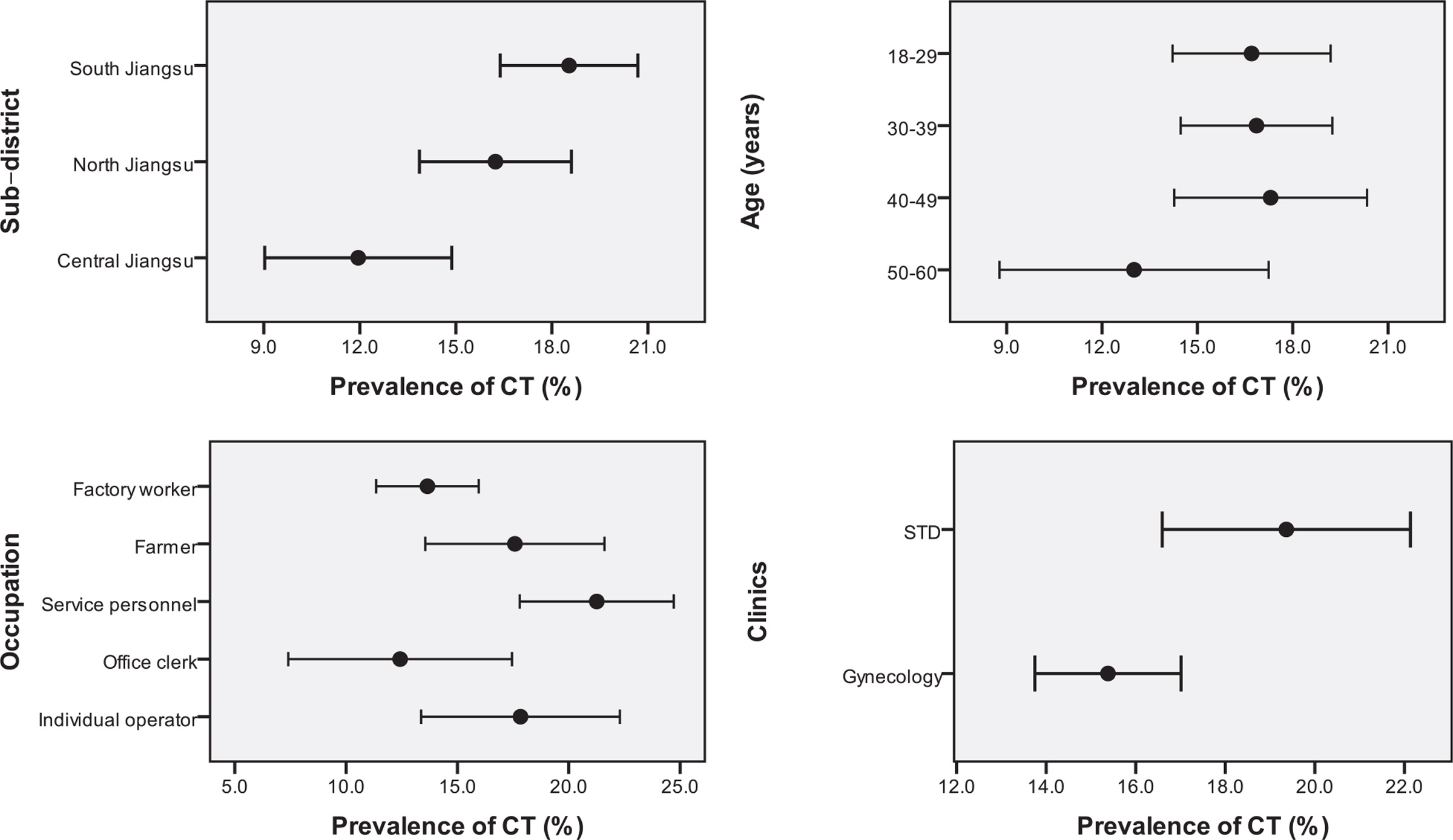
Differences in the prevalence of Chlamydia trachomatis (CT) infection between subgroups of 4 main variables
Through univariate and multivariate analysis, female outpatients from STD clinics (19.4%) and South Jiangsu (18.5%) were higher. Female outpatients who were service personnel (21.3%) or farmers (17.6%), lived in South Jiangsu (18.5%), and were from STD clinics (19.4%) were more potential to be infected with CT.
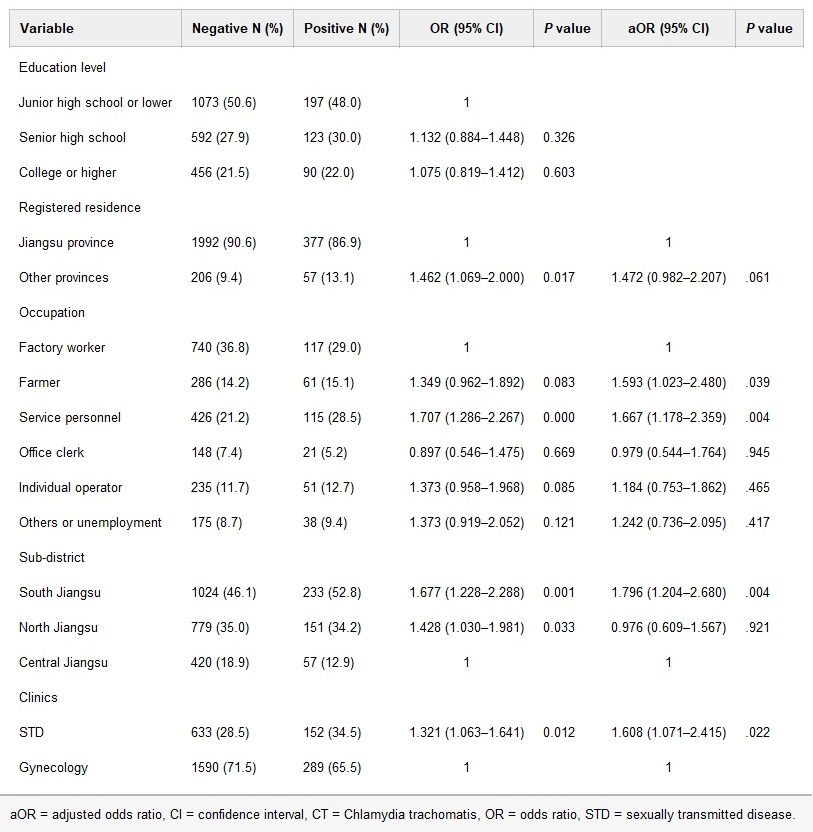
Univariate and multivariate analysis of factors associated with CT infection among women attending STD and gynecology clinics
This study indicates a high prevalence of CT infection among women in Jiangsu province, China. According to the CDC, untreated chlamydia may also increase the chances of getting or giving HIV, highlighting the importance of CT screening on the chlamydia control and prevention agenda.
Bioperfectus Chlamydia Trachomatis Real-Time PCR Kit was honored to be included in this study. Launched to international market in August 2021, the kit has received CE approval and NMPA certificate and entered over 100 countries. Samples can be obtained from a clinical male urethral swab, female endocervical swab, or urine. It is much more convenient to collect urine samples, especially when screening for more populations.
Bioperfectus provide Total STDs PCR Solution with Nucleic Acid Extraction Platform, STD Real-time PCR Kits and Real-time PCR System to satisfy various testing scenarios including general screening and screening for risk groups with high sensitivity and specificity to ensure reliable performance in clinical practice.
Followed by the release of “One Solution, One Supplier, One Response”, once an abnormal result occurs, the problem will be immediately solved by just one call to our customer service team as Bioperfectus provides comprehensive solution from sample preparation to nucleic acid amplification to our global partners and customers.

References

